EXCLUSIVE: Leaked AP Chem Equation Sheet That Teachers Are Trying To Hide!
Are you struggling to memorize every formula and constant for your upcoming AP Chemistry exam? What if we told you there's a comprehensive reference sheet that could save you countless hours of study time? The AP Chemistry equation sheet has been a game-changer for students, yet many aren't fully utilizing its potential. In this exclusive breakdown, we'll reveal everything you need to know about this powerful tool that some teachers might not want you to fully understand!
Understanding the AP Chemistry Formula Sheet
The AP Chemistry formula sheet is much more than just a collection of equations—it's your secret weapon for exam success. This comprehensive document provides a concise and organized compilation of essential equations, constants, and other pertinent information that students need to master for the exam. Understanding how to navigate this resource effectively can significantly impact your performance.
The equation sheet is designed to be user-friendly, with formulas organized by topic area for quick reference. You'll find everything from basic gas laws to complex thermodynamic equations, all in one place. The sheet typically includes standard reduction potentials, equilibrium constants, and common conversion factors that you'll need throughout the exam. What makes it truly valuable is that it's provided to you during the actual test, meaning you don't have to memorize every single formula—you just need to know how to use them correctly.
- The Porn Industrys Viral Hack For Gel X Nail Removal That Will Blow Your Mind
- The Porn Leak That Changed Everything Chers True Age Finally Exposed
- You Wont Believe What Was Leaked From Walmart St Croix Falls Back Room
One common misconception is that having the formula sheet means you don't need to study. This couldn't be further from the truth! The sheet is a reference tool, not a replacement for understanding. You still need to comprehend the underlying concepts and know which formula to apply in different scenarios. Think of it as having a calculator—it's an incredibly useful tool, but only if you understand the mathematical principles behind it.
What's Included in the 2025 AP Chemistry Equation Sheet
The 2025 AP Chemistry equation sheet covers key concepts across nine units, including atomic structure, bonding, chemical reactions, kinetics, thermodynamics, equilibrium, acids and bases, and applications of thermodynamics. This comprehensive coverage ensures you have access to every formula you might need during the exam. The sheet is carefully curated to include only the most essential equations, eliminating unnecessary clutter while providing everything required for success.
For atomic structure, you'll find equations related to electron configuration, quantum numbers, and periodic trends. The bonding section includes formulas for molecular geometry, hybridization, and intermolecular forces. Chemical reactions cover stoichiometry, limiting reactants, and percent yield calculations. Kinetics includes rate laws, integrated rate equations, and half-life formulas. Thermodynamics encompasses enthalpy, entropy, and Gibbs free energy equations. Equilibrium provides formulas for equilibrium constants, ICE tables, and Le Chatelier's principle applications.
- Ice Spices Body Transformation Exposed The Leaked Diet Plan That Changed Everything
- Nude Truth Revealed Why Premium Outlet Tucson Az Is The Most Controversial Mall In America
- Ramon Rodriguez Sex Tape Scandal Shocking New Leak Exposes Everything
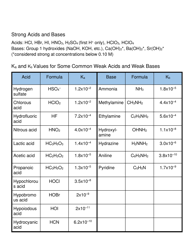
The acids and bases section includes pH calculations, Ka and Kb expressions, and buffer equations. Applications of thermodynamics cover electrochemistry, including cell potential calculations and the Nernst equation. Throughout the sheet, you'll also find important constants like Avogadro's number, the gas constant (R) in various units, and standard reduction potentials for common half-reactions. These constants are crucial for calculations and are provided to save you from memorizing numerous values.
How to Effectively Use the Equation Sheet
Mastering the AP Chemistry equation sheet isn't just about having it during the exam—it's about knowing how to use it efficiently under time pressure. Start by familiarizing yourself with the layout before test day. Spend time during your study sessions using the sheet as you would during the actual exam. This practice will help you develop muscle memory for where specific formulas are located, saving precious seconds when it matters most.
When approaching a problem, first identify what type of calculation you need to perform, then quickly locate the relevant section of the equation sheet. For example, if you're dealing with gas laws, you'll know to look in the gases section rather than flipping through the entire sheet. This strategic approach can significantly reduce the time spent searching for formulas. Remember, every second counts during the AP Chemistry exam!
Another crucial aspect of using the equation sheet effectively is understanding the units associated with each formula. Many students make the mistake of plugging in values without considering unit conversions, leading to incorrect answers. Pay special attention to the units of constants and ensure your given values are in compatible units before performing calculations. For instance, when using the gas constant R, be aware that it comes in different forms (0.0821 L·atm/mol·K, 8.314 J/mol·K, etc.), and you must use the version that matches your other units.
Key Formulas You Must Master
While the equation sheet provides all the formulas you'll need, certain equations deserve special attention due to their frequent appearance on the exam. The ideal gas law (PV = nRT) is fundamental and appears in various forms throughout the test. You should be comfortable rearranging this equation to solve for any variable and understand how it relates to other gas laws like Boyle's Law, Charles's Law, and Avogadro's Law.
Thermodynamic equations are another critical area. The Gibbs free energy equation (ΔG = ΔH - TΔS) is essential for determining reaction spontaneity. You should also master the relationship between ΔG and the equilibrium constant (ΔG° = -RT ln K). These equations often appear together in free-response questions, requiring you to connect multiple concepts. Additionally, the Nernst equation for calculating cell potential under non-standard conditions is crucial for electrochemistry problems.
For kinetics, the rate law expressions and integrated rate equations for zero, first, and second-order reactions are must-know formulas. You should be able to determine reaction order from experimental data and calculate half-lives for different reaction orders. In equilibrium, the expression for the equilibrium constant (K = [products]/[reactants]) and its relationship to reaction quotient Q are fundamental. Understanding how to use ICE tables to solve equilibrium problems is also essential for success.
Mnemonics and Memory Aids
Even with the equation sheet, having certain formulas memorized can save you valuable time during the exam. Creating mnemonics can help you remember complex equations and concepts more easily. For example, to remember the quadratic formula (which occasionally appears in equilibrium problems), you might use the phrase "Negative Boy was always sad and cried" to recall the negative, b, plus/minus, square root, b², four, a, c structure.
For thermodynamics, the phrase "Good Honey Tastes Sweet" can help you remember that Gibbs free energy (ΔG) equals enthalpy (ΔH) minus temperature (T) times entropy (ΔS). In electrochemistry, remembering that "Red Cat" (reduction occurs at the cathode) and "An Ox" (oxidation occurs at the anode) can help you quickly identify electrode processes. Creating your own mnemonics based on your interests or experiences can make them even more memorable and effective.
Beyond mnemonics, understanding the conceptual basis behind formulas can improve your recall and application. For instance, rather than just memorizing PV = nRT, understand that pressure and volume are inversely related when temperature and moles are constant (Boyle's Law). This conceptual understanding allows you to derive related equations even if you momentarily forget a specific formula. Focus on understanding the "why" behind each equation, not just the "what."
Practice Makes Perfect
The most effective way to master the AP Chemistry equation sheet is through consistent practice with real exam questions. The College Board provides numerous past exam questions that you can use to familiarize yourself with how formulas are applied in different contexts. Start by working through problems without the equation sheet to identify which formulas you need to reference, then gradually incorporate the sheet as you would during the actual exam.
When practicing, pay attention to the types of questions that frequently appear. For example, gas law problems often involve calculating molar mass from density measurements, while thermodynamics questions frequently ask you to determine whether reactions are spontaneous. By recognizing these patterns, you can develop a strategic approach to the exam, knowing which sections of the equation sheet to reference for different question types.
Time management is crucial during practice sessions. Set a timer to simulate exam conditions and practice locating formulas quickly. You might discover that certain sections of the equation sheet are more frequently used than others, allowing you to develop a mental map of the most important areas. Additionally, practice showing your work clearly, as partial credit is often awarded on free-response questions even if the final answer is incorrect.
Common Mistakes to Avoid
Even with the equation sheet, students often make common mistakes that cost them valuable points. One frequent error is failing to properly convert units before plugging values into equations. For example, using pressure in atm with a gas constant R in J/(mol·K) will give an incorrect answer. Always double-check that your units are compatible with the constants you're using.
Another common mistake is misapplying formulas or using the wrong equation for a given situation. For instance, using the ideal gas law when dealing with real gases under extreme conditions, or applying the wrong rate law expression for a given reaction order. To avoid these errors, carefully read each question and identify keywords that indicate which concept or equation is being tested. Look for clues like "at equilibrium," "rate of reaction," or "spontaneity" to guide your approach.
Many students also struggle with significant figures and rounding errors. The AP Chemistry exam typically requires answers to be reported with the correct number of significant figures, and premature rounding can lead to incorrect final answers. Keep extra significant figures during intermediate calculations and only round your final answer. Additionally, pay attention to whether the question asks for a specific unit or format for your answer.
Expert Tips for Exam Day
On the day of the AP Chemistry exam, having a strategic approach to using the equation sheet can give you a significant advantage. First, quickly scan through the entire sheet when you receive it to refresh your memory of what's available. This quick overview can help you identify any formulas you might have forgotten to review. Then, as you work through the exam, use the sheet methodically, referring to specific sections rather than flipping through randomly.
For multiple-choice questions, try to solve the problem on your own first, then use the equation sheet to verify your approach or find the exact formula you need. This prevents you from becoming overly reliant on the sheet and helps you work more efficiently. For free-response questions, clearly show your setup and the formulas you're using, even if you have to reference the equation sheet. This demonstrates your understanding of the problem-solving process and can earn you partial credit.
Remember that the equation sheet is provided for all sections of the exam, including the multiple-choice portion. Don't hesitate to use it whenever you're unsure about a formula or need to verify a constant. However, be mindful of time—if you find yourself spending too long searching for a specific equation, it might be better to make an educated guess and move on. Time management is crucial, so use the sheet strategically rather than exhaustively.
Conclusion
The AP Chemistry equation sheet is an invaluable resource that, when used effectively, can significantly boost your exam performance. It's not just a collection of formulas—it's a comprehensive reference tool that covers every key concept you'll need to master for success. By understanding its layout, practicing with it regularly, and developing strategic approaches to different question types, you can maximize its potential and minimize exam-day stress.
Remember that the equation sheet is designed to complement your knowledge, not replace it. Your understanding of chemical principles, problem-solving skills, and ability to apply concepts in different contexts are what truly matter. Use the sheet as a safety net and time-saver, but continue to build your conceptual foundation through thorough studying and practice.
As you prepare for your AP Chemistry exam, make the equation sheet an integral part of your study routine. Familiarize yourself with its contents, practice using it under timed conditions, and develop confidence in your ability to navigate it efficiently. With the right preparation and strategic use of this powerful tool, you'll be well-equipped to tackle any challenge the exam presents and achieve the score you're aiming for.